Progressing treatments in cancer immunotherapy clinical trials
Four publications outline how mass cytometry can shed light on mechanism of action, predictive response and relevant biomarkers across an array of therapeutic areas

Identifying and quantifying cell types in patient samples allows a look at the cell diversity and changes in proportion of cell types among different samples. These specific and precise adjustments made by the immune system can expose the impact of a disease on the body or a treatment on the disease. Yet, finding these potentially small but relevant correlations can be a challenge when sifting through millions of cells at once. Further, the ability to measure functional states and signatures of cells is key to understanding immune responses to disease and treatment
Here, we highlight the ability of tools that assess intracellular activity to help simultaneously determine cell type and function, generating quantitative data for all cell populations in a sample. The four articles below outline how, through wide coverage of surface and functional markers, mass cytometry can help reveal mechanism of action, predictive response and relevant biomarkers across a variety of therapeutic areas, helping improve immunotherapies through mitigating risks of off-target effects and understanding patient stratification and immune-response biomarkers.
New biomarkers to stratify patients into non-progressors and progressors
A Phase 2 clinical trial (NCT03367741) as described in Lheureux et al. focused on combining a checkpoint inhibitor (nivolumab) and an antiangiogenic agent (cabozantinib) in an effort to treat endometrial cancer (EC). Women with advanced, recurrent or metastatic EC were given either nivolumab with cabozantinib or nivolumab alone; the trial was designed to detect differences in progression-free survival (PFS) between these two groups. A 36-marker CyTOF™ panel was used to analyze 40 baseline biopsies across the treatment arms and identified 35 unique immune cell subsets constituting the major immune cell populations. Mass cytometry was critical in identifying subsets of immune cells that enabled researchers to stratify patients into non-progressors and progressors, becoming the first reported biomarkers for response to immunotherapy in EC.
This groundbreaking research, which the authors say is the first study demonstrating benefit from cabozantinib-nivolumab combination therapy in patients with heavily pretreated recurrent EC, confirmed that PFS was improved with combination therapy vs. treatment with only nivolumab, emphasizing the benefit of combining antiangiogenic agents and immuno-oncology.
“CyTOF analysis of fresh baseline biopsies provides the first high-dimensional insight into the immune microenvironment of recurrent EC.”
High-parameter analysis identifies immune predictors of improved response rate
Amid a growing interest in identifying predictive biomarkers of immune checkpoint inhibitor (ICI) efficacy in non-small-cell lung cancer (NSCLC), a publication by Rochigneux et al. evaluated circulating immune predictors of pembrolizumab efficacy in patients with advanced NSCLC.
The researchers established a retrospective cohort of patients with advanced NSCLC enrolled at the University of California Los Angeles (UCLA) on the KEYNOTE-001 (NCT01295827) Phase 1 trial. CyTOF technology was used to perform deep phenotyping of multiple immune cell types at once in baseline samples from 27 pembrolizumab-treated patients and identified populations in peripheral blood that predicted pembrolizumab efficacy.
The team demonstrated that blood baseline frequencies of classical monocytes, NK cells and ICOS+ CD4+ T cells are associated with improved pembrolizumab efficacy (objective response rates, PFS and overall survival); they also reported that a baseline immune peripheral score combining these three populations strongly predicts pembrolizumab efficacy. These findings may improve prediction of ICI benefit in patients with advanced NSCLC.
“Recently, the advent of CyTOF has enabled high-dimensional and unbiased examination of the immune system, allowing simultaneous interrogation of a large number of parameters. We hypothesized that CyTOF analysis of blood immune cells before immunotherapy could identify new predictors of ICI efficacy.”
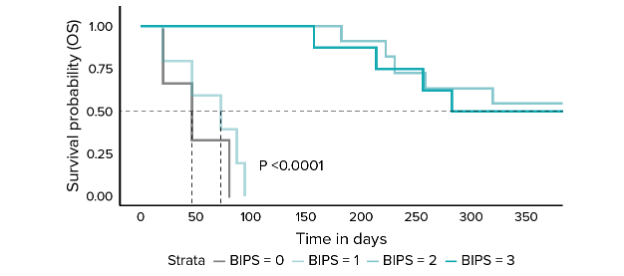
CyTOF analysis demonstrates that blood baseline frequencies of classical monocytes, NK cells and ICOS+ CD4+ T cells are significantly associated with improved objective response rates, progression-free survival and overall survival (shown here). BIPS = a simple score developed by adding one point when the baseline frequency of each cell population was over the pre-cited threshold. BIPS values range from 0-3, with 0-1 identifying progressive disease and 2–3 identifying patients with clinical benefit from treatment.
The association between specific cell subtypes and pathologic response in melanoma
As part of NeoACTIVATE (NCT03554083), a Phase 2 trial, patients with clinically evident resectable stage 3 melanoma were assessed to evaluate the efficacy and tolerability of neoadjuvant combinatorial targeted therapy and immunotherapy.
Thirty patients received either neoadjuvant vemurafenib, cobimetinib and atezolizumab combination therapy or cobimetinib and atezolizumab combination therapy. Hieken et al. reported on the neoadjuvant findings, hypothesizing that a combination of targeted therapy and immunotherapy would drive robust pathologic responses.
CyTOF technology was used to assess the frequencies of relevant immune cell populations in PBMC at four key timepoints, revealing associations between specific cell subtypes and pathologic response that other technologies could not find. For example, the data showed an increase in the frequency of CD8+ central memory T cells (TCM) present at baseline in responding patients. This result provides insight into new treatment regimens and suggests CD8+ TCM cell activity, known for playing a role in disease eradication in lymph nodes, is important for response to therapy.
“Since we saw an association between CD8+ T cells and pathologic response, we also [used mass cytometry to assess] the frequencies of CD8+ naive T cells, CD8+ central memory T cells (TCM), CD8+ effector memory T cells (TEM), and CD8+ effector memory T cells re-expressing CD45RA (TEMRA). Notably, increased baseline TCM were observed in patients with favorable versus unfavorable pathologic responses.”
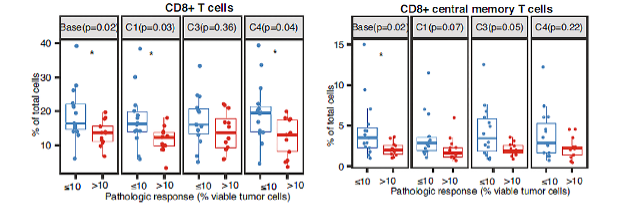
CyTOF technology quantified cell subsets at baseline, after treatment cycle 1 (C1), after completion of neoadjuvant treatment (C3) and after operation (C4) across both cohorts. Frequencies of CD8+ T cells as a percentage of total PBMC are grouped by response status. Increased baseline CD8+ TCM cells were observed in patients with favorable versus unfavorable pathologic responses.
Understanding mechanisms of action in CAR therapies
NKG2D ligands are a promising target for CAR T cell therapy treating acute myeloid leukemia (AML) and T cell acute lymphoblastic leukemia (T-ALL). In a follow-on from the first-in human Phase 1 study of NKG2D CAR T cells in patients with AML and multiple myeloma, Driouk at el. provide comprehensive preclinical evidence of in vitro NKG2D CAR T cell efficacy in T-ALL and AML and demonstrate that HDAC inhibition achieves selective upregulation of NKG2D ligands on the surface of AML blasts, which leads to enhanced NKG2D CAR T cell efficacy.
CyTOF technology was used to evaluate the characteristics of NKG2D CAR T cells when encountering NKG2D ligands on tumor cells and revealed a phenotypically heterogeneous population of CAR T cells with a predominance of effector memory phenotype (TEM) and central memory (TCM) populations. Distinct populations of stem cell memory (TSCM) and terminal effector memory (TEMRA) populations were also present.
Mass cytometry was integral in revealing the mechanism of NKG2D CAR T cell activity: NKG2D CAR T cells showed significant upregulation of ICOS, CD25, 41BB and OX-40. A similar upregulation of PD-1 and LAG-3 suggests that NKG2D CAR T cell response could be further enhanced in combination with checkpoint blockade or genetic modification strategies. A decrease in Ki-67 and CXCR3 was seen in TEM and TEMRA CAR subsets and was maintained in TSCM and TCM subsets responsible for an ongoing NKG2D CAR T cell response.
